Introduction
Traceability isn’t just a technology—it’s a regulatory reality that’s shaping how industries operate. From food and pharma to electronics and packaging, global mandates are driving digital change.
Understanding these regulations—and turning them into opportunities—can set your business apart.
The Shifting Regulatory Landscape
US: FDA’s FSMA and the Drug Supply Chain Security Act (DSCSA) for pharma.
EU: EU Digital Product Passport (DPP) and FMD serialization rules for pharmaceuticals.
Asia: China’s UDI for medical devices and strong food traceability measures.

Impact Across Industries
These rules are more than red tape—they’re about consumer safety, sustainability, and unlocking new markets. Compliance is becoming a ticket to trade and a marker of brand trust.
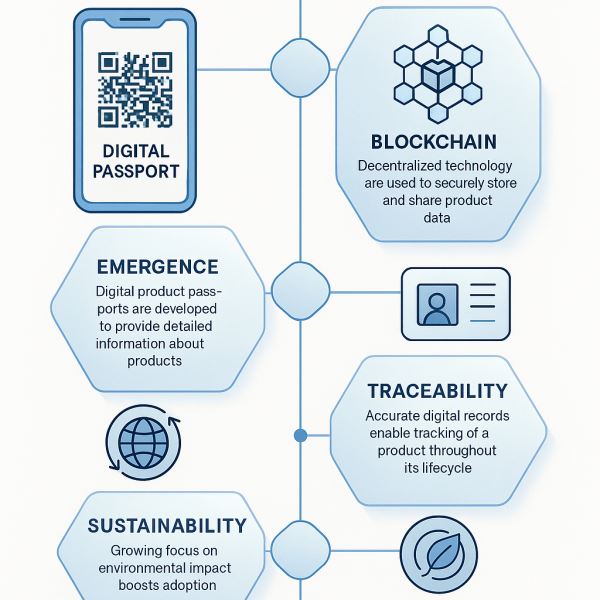
Future: Digital Product Passports and the Circular Economy
The EU’s Digital Product Passport is a sign of things to come: transparency, sustainability, and lifecycle data. Businesses that embrace these changes will lead the way in the next decade.
Case Studies: Compliance in Action
Nestlé: Proactive traceability to meet FSMA and EU DPP.
Roche: Compliance-driven pharma serialization.
Adidas: Circular economy packaging standards met with smart tracking.

Strategic Takeaways
Map your global compliance requirements.
Invest in digital platforms that adapt to evolving mandates.
Use compliance as a competitive differentiator, not just a box-ticking exercise.
Conclusion
Regulatory traceability isn’t a burden—it’s a chance to lead in quality, safety, and sustainability. Businesses that embrace these changes today will define tomorrow’s markets.

