Introduction
Serialization and traceability are often used interchangeably, but they are not the same. In fact, understanding the difference can mean the difference between basic compliance and full supply chain intelligence.
Whether you’re in pharma, food, electronics, or consumer goods, it’s critical to know what each term means, where it applies, and how to integrate both for maximum benefit.
What is Serialization?
Serialization is the process of assigning a unique identifier to each unit of a product. These identifiers are usually applied at the packaging level using 2D barcodes, QR codes, or RFID tags.
A serialized code might include: – Product ID
– Batch or lot number
– Serial number
– Expiry date
– Manufacturing location
Serialization is most commonly used in industries where unit-level verification is needed, such as: – Pharmaceuticals (EU FMD, DSCSA) – Electronics – Luxury goods
 What is Traceability?
What is Traceability?
Traceability refers to the ability to track and trace the entire lifecycle of a product, from raw material to end consumer and beyond.
It includes: – Batch-level data
– Processing steps and timestamps
– Handling, storage, and transportation logs
– Compliance events and quality inspections
Traceability is broader than serialization and can exist without unit-level codes. It’s essential in: – Food safety (FSMA, HACCP) – Textiles and garments – Industrial manufacturing
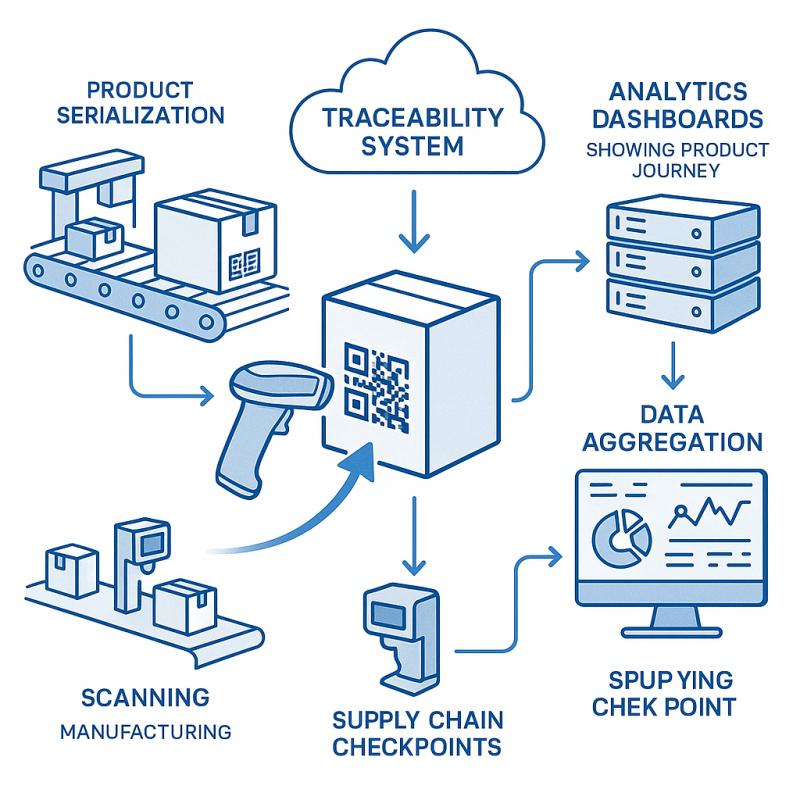 How They Complement Each Other
How They Complement Each Other
Serialization enables precise identification, but without a traceability system, it lacks context. Traceability connects the dots across: – Production batches – Quality control – Distribution events – Recall management
Think of serialization as the “what”, and traceability as the “where, when, how, and why.”

Real-World Use Cases
- Pharma: Serialization ensures every pack is authentic. Traceability logs temperature during storage and shipment. Together, they enable compliance and patient safety.
- Food: Batch-level traceability handles recalls. Serialization can validate individual product freshness or allergen data for end users.
- Electronics: Serialized codes are used to prevent counterfeiting; traceability helps track component sourcing and warranty claims.
Strategic Takeaways
- Serialization is a subset of traceability: It gives visibility at the unit level.
- Use both where possible: Especially in compliance-heavy or high-risk industries.
- Choose your tech wisely: QR for consumer goods, RFID for bulk, 2D DataMatrix for pharma.
- Train your teams: Success depends on accurate data capture and system integration.

Conclusion
While serialization assigns identity, traceability delivers insight. Together, they form the foundation of transparent, accountable, and intelligent supply chains.
Don’t just serialize to comply—trace to lead.

