Introduction
Fake medicines are a global crisis. According to the WHO, 1 in 10 medical products in low- and middle-income countries is substandard or falsified.
The result? Lives lost, trust broken, and public health undermined.
But thanks to serialization and digital traceability, the pharmaceutical industry is now equipped with powerful tools to fight back.
Serialization: A Unique Identity for Every Pack
Serialization involves printing a unique 2D barcode on each saleable unit of medicine, containing: – Global Trade Item Number (GTIN) – Serial number – Batch or lot number – Expiry date
This makes every pack traceable, verifiable, and—if needed—recallable.
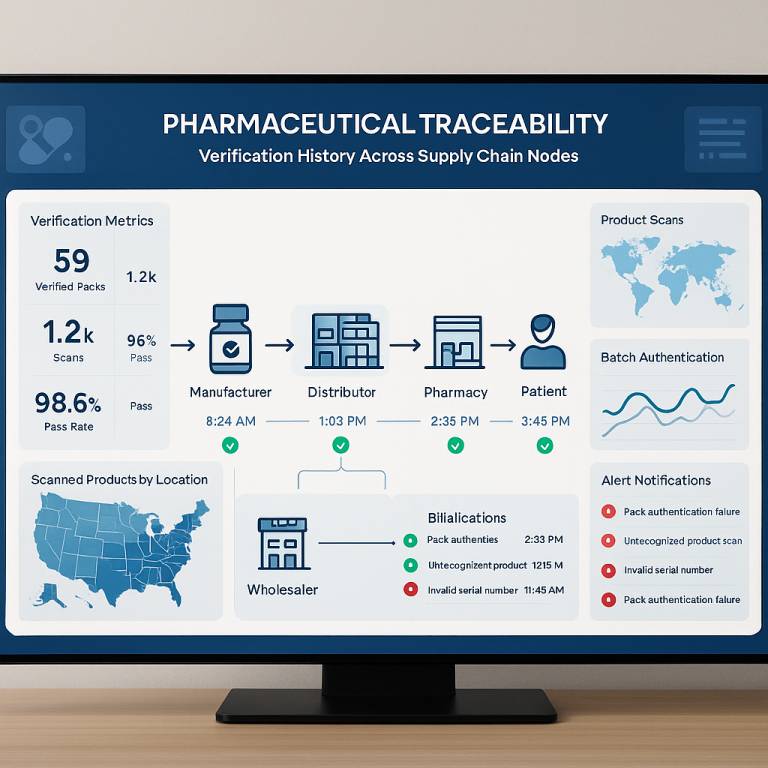
How Traceability Prevents Counterfeiting
Authentication at Every Level Distributors, pharmacists, and even patients can scan the pack using mobile apps or systems to verify its authenticity in real-time.
Tamper-Proof Supply Chain Traceability systems log each movement across the supply chain, making it nearly impossible for fakes to enter unnoticed.
Instant Recalls If a defect is discovered, traceability allows authorities to isolate and recall only the affected packs, not the entire batch.
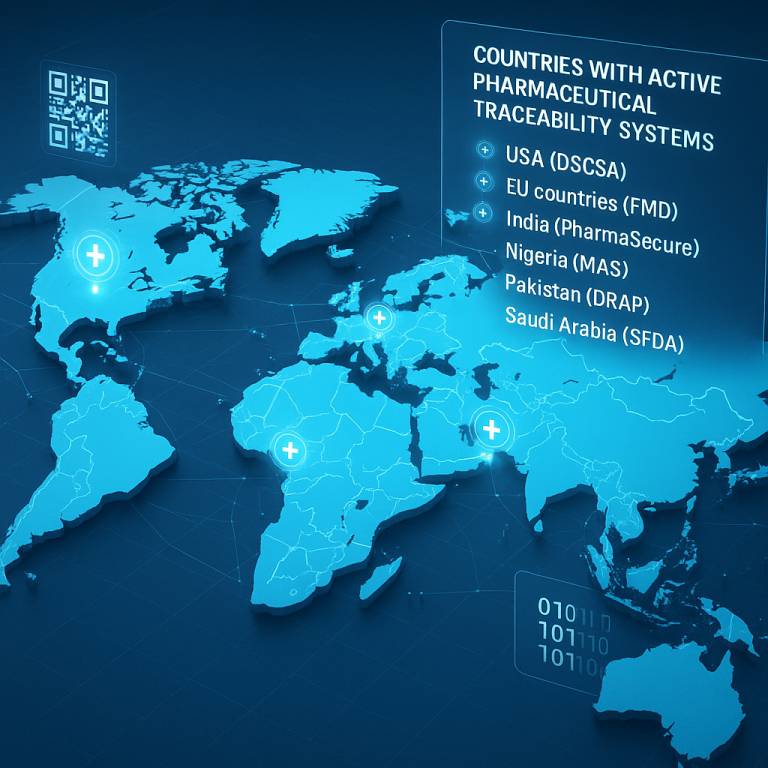
 Global Standards & National Systems
Global Standards & National Systems
- DSCSA (USA): Requires full unit-level serialization and verification across the pharmaceutical supply chain.
- EU FMD: Includes serialization, tamper-evident packaging, and centralized verification systems.
- DRAP (Pakistan): Mandates serialization and GS1 standards for medicine manufacturers.
- SFDA (Saudi Arabia): Uses a national Drug Track & Trace System with real-time alerts.
 Real-World Success Stories
Real-World Success Stories
- India’s Pharma Secure program helped consumers verify over 1 billion medicine packs.
- Pfizer has used serialization to identify and stop parallel trading and counterfeit injections in several regions.
- Nigeria’s MAS (Mobile Authentication Service) allowed patients to verify medicines via SMS, reducing fake drug cases.
 Strategic Takeaways
Strategic Takeaways
- Implement serialization from the start of production
- Integrate traceability into packaging and distribution workflows
- Train pharmacies and regulators to scan and verify units
- Use consumer-facing apps to build public trust and safety
Conclusion
Traceability is not just about compliance in pharma. It’s about saving lives.
With the right systems in place, manufacturers can ensure that every medicine pack that reaches a patient is real, safe, and effective.
Because no patient should ever have to question the medicine in their hand.

