Introduction
When it comes to medicine, traceability isn’t optional—it’s lifesaving. Around the world, pharmaceutical serialization laws are transforming how drugs are tracked, verified, and recalled.
From the US to the EU to emerging markets, serialization ensures that every pack of medicine is traceable, authentic, and compliant.
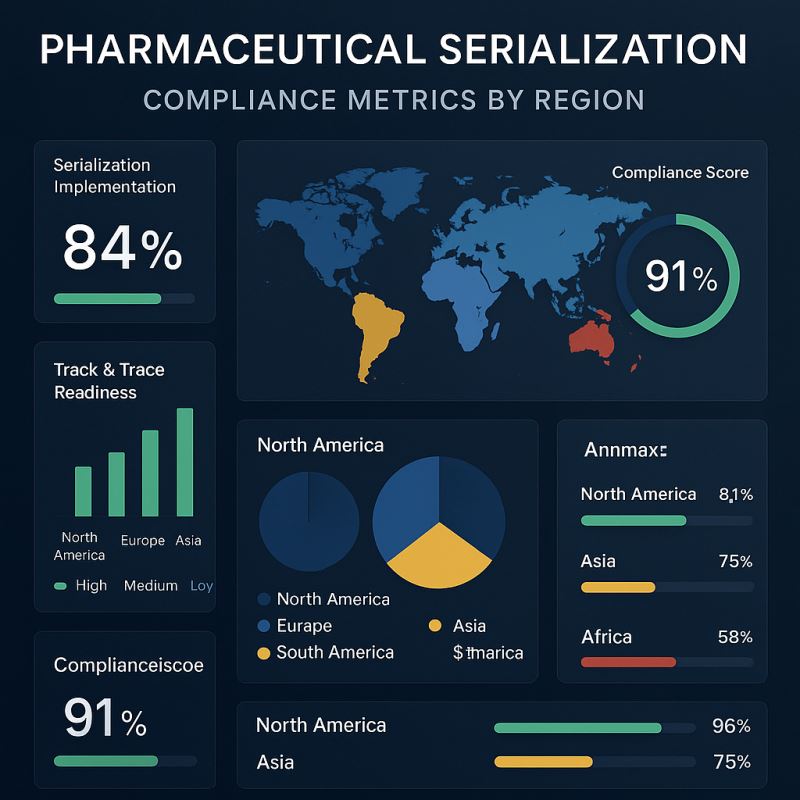 The Global Serialization Landscape
The Global Serialization Landscape
United States – DSCSA (Drug Supply Chain Security Act)
Requires complete unit-level tracking of prescription drugs by 2024—serialization, verification, and data exchange.
Europe – Falsified Medicines Directive (FMD)
Mandates 2D barcodes, tamper-evident packaging, and national databases for serialized medicine tracking.
Asia – China’s Drug Administration Law & India’s Barcode Mandate
Rising regulations around serialization and electronic tracking of exports.
How Serialization Supports Pharma Safety
- Recall Precision: Instantly isolate faulty batches using serialized IDs.
- Anti-Counterfeiting: Detect and eliminate fake drugs in the supply chain.
- Regulatory Compliance: Automate recordkeeping, reporting, and inspection readiness.
- Global Trade Access: Meet import/export standards across major markets.
Tech Stack for Serialization
- 2D DataMatrix barcodes for global scanning standards
- Cloud traceability platforms for centralized data access
- RFID and blockchain for added security and auditing
- Pharmacy-level verification systems to ensure authenticity at dispense point
Brand Case Studies
- Novartis uses blockchain for global batch traceability.
- Pfizer integrates serialization into cold-chain tracking.
- GSK links serialization to global distribution and recall dashboards.
Strategic Takeaways
- Evaluate serialization readiness across all SKUs and markets.
- Train stakeholders—serialization is only effective when universally adopted.
- Combine serialization with anti-tampering packaging for maximum trust.

Conclusion
Pharma serialization is more than a regulation—it’s a promise. A promise of safety, quality, and global readiness. The companies that master serialization today will lead the industry tomorrow.

